This now five-part case study focuses on improving customer satisfaction at two of a company’s diagnostic clinics – Centers A and B. In Part One, the company worked at Center A to reduce patient turnaround time, a defining component of patient satisfaction. In Part Two, the chain’s improvement story focuses on increasing patient delight at Center B. In Part Three, the story returned to improving the processes of the lab in Center A. In Part Four, turnaround time in a new centralized laboratory is improved. Check back next Monday for Part Five.
A chain of medical diagnostic clinics was developed from the ground up. After two years of hectic expansion marked by acquisitions and setting up greenfield clinics across a number of cities, it became clear that due to the different inherited processes across those acquisitions, the chain needed to focus more on improving and standardizing the quality of service offered, and improving its customer satisfaction rankings.
After successfully improving the processes in laboratory of Center A, the chain of medical dianostic clinics wanted to move forward with improving the turnaround time (TAT) in the newly established centralized laboratory servicing the 62 individual lab centers.
A cross-functional team was set up to undertake the project.
Define the Problem
The group brainstormed a list of 52 problems related to the laboratory (see Appendix 1). Discussions revealed that most of the problems listed were causes of problems rather than problems in and of themselves. There were only four “real” problems:
- Delay (i.e., TAT)
- Quality
- Safety
- Service
These were prioritized to select the “vital” problem using a weighted average table. Scores indicated that delay (i.e., TAT) was the vital problem. The next step was to measure the problem.
Turnaround problems can be resolved using just-in-time (JIT) techniques. Here, this involved deciding at the outset the start and end points of the process for which the TAT was to be reduced.
Complete TAT starts with the customer arriving at the Diagnostic Center and ends with the report being given to them. The overall desire of senior management was that customers visiting the Center in the morning should receive their reports the same day and customers visiting in the afternoon should receive their reports the next morning.
The 30,000-foot-level process steps were listed and are shown in Table 1. Also shown are the various start and end points of the project that could be followed.
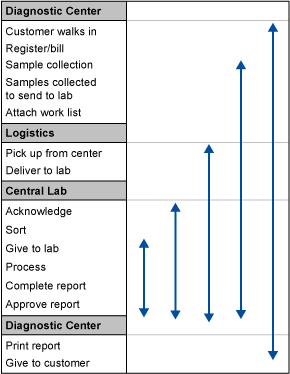
To select which part of the chain should be addressed, data was required for where the maximum time was being spent. A format was prepared to collect data for each part of the chain by tracking 100 samples as shown in Table 2.
| Table 2: Excerpt of TAT Data Collection from Customer Registration to Report Handover | ||||
| Responsible Division | Dept. | Item | Customer 1 | Customer 2 |
| Date | January 7, 2012 | January 7, 2012 | ||
| Customer ID | 112608 | 112614 | ||
| Customer name | X | Y | ||
| Diagnostic center | Front desk admin | Registration time | 07:42 | 08:15 |
| Diagnostic center | Lab | Sample collection time | 08:03 | 08:37 |
| Lab | Sample ready for dispatch | 08:45 | 08:45 | |
| Back-end admin | Logistics | Sample pickup time | 09:00 | 09:00 |
| Back-end admin | Logistics | Sample delivered time | Varies | Varies |
| Central lab | Report upload time | 17:39 | 15:17 | |
| Diagnostic center | Front desk admin | Report printing time | 17:47 | 15:42 |
| Diagnostic center | Lab | Report dispatch to counter | 17:55 | 15:55 |
| TAT (hours:minutes) | 10:13 | 07:40 | ||
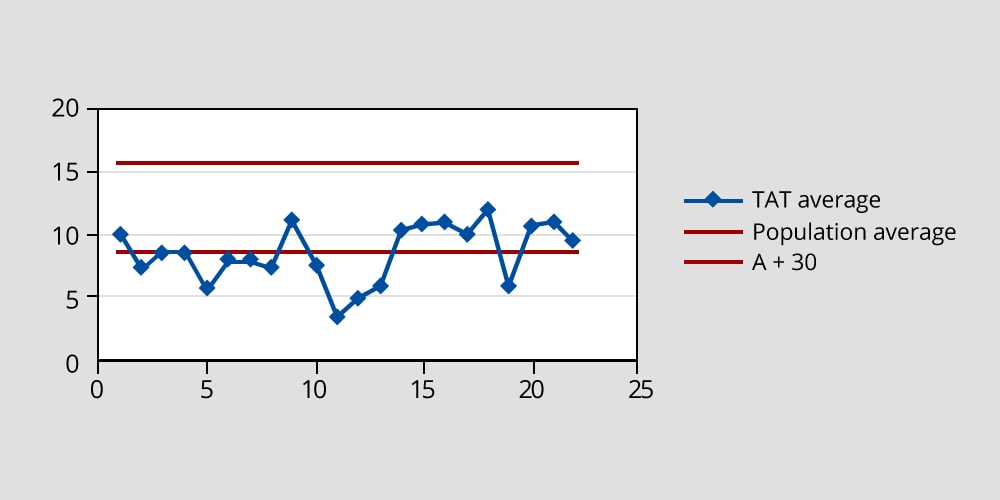
The concept of sigma was introduced to the team to measure variability in addition to the average. The average and sigma for the current state TAT was calculated as follows.
Average = 15.9 hours
Average + 3σ = 56 hours
The logistics time was found to be 2.5 hours to 3 hours within the 15.9 hours average. The team set an objective of reducing the average TAT by 50 percent – from 15.9 hours to 8 hours.
A control chart was developed to track the TAT. The initial chart is shown in the figure below.
Mapping the Current State
Another metric was also introduced – the percentage of reports completed/returned to patients in a 24-hour period of time. Existing data showed that this was at 76 percent.
The next step was mapping the current state process, by looking at:
- Volume to be processed
- Arrival pattern
- Time to process the smallest unit at each stage of the process
- Deployment pattern of people, equipment
From there, the future state process map would be developed while applying the following techniques to reduce work in process (i.e., waiting time) and, therefore, TAT:
- Reducing non-value adding stages
- Balancing the line (i.e., ensure capacity available can process arrivals at the required speed)
The volumes to be processed were based upon the Center’s past data and growth projections. The target volume to be processed was 50,000 tests per day. Since different tests require different equipment and times, it was necessary to ascertain the ratios of the tests required to be able to make all of them flow (Table 3) based on 25 working days per month.
| Table 3: Volume Estimates for Central Lab | |
| Volume | Per Day |
| Patients | 300 |
| Samples | 650-700 |
| Tests | 1,200 |
| Department | Per Day |
| Blood | 76% |
| Urine | 19% |
| Others | 5% |
Arrival Patterns
Arrival patterns were studied by the hour for four days – for all samples, with blood and urine samples separately (the most common tests and the majority of tests ordered; see details in Appendix 2) – and are shown in Table 4. All patterns are based on 12-hour increments in time.
| Table 4: Summary of Arrival Patterns of Samples | |||
| Per Hour | All Samples | Blood | Urine |
| Average | 65 | 55 | 9 |
| Peak | 136 | 80-100 | 33 |
| Average Per Day | 500-600 | 100-200 | |
Testing Capacity
Next, it was time to look at testing capacity. A list of all equipment was prepared according to section (e.g., biochemistry, hematology, etc.). Against each test the information shown below was assessed:
- Machine(s)
- Batch (including batch size) or individual
- Time
- Units
- Speed
- Barcode logging of samples
- Reports automatically generated
- Printout of reports
Equipment Capacity
The results of an equipment capacity analysis helped explain the processes, the capacity, degree of automation/manual work involved and the batch/flow nature of the work.
- Average time per test: A comprehensive list of tests was prepared and the average time per test was recorded. (See Appendix 3.)
- Manpower deployment: When the arrival pattern of the load to be processed required deploying personnel to best match the incoming load patterns, manpower deployment was critical and also mapped.
- Improving the TAT: Performed section by section.
Improved TAT Process
The following process was used to develop the improved TAT process:
- Map key stages of process
- Determine stations
- Smallest unit though line at each station
- Decide batch size
- Time/unit at each station
- Ensure flow capability at each station: capacity to process load-in time required
- Instruct team
- Run one batch without waiting
- Record lead time/waiting time/non-value added times
- Kill problems to smooth workflow
- Try longer and longer runs at higher and higher loads
A green channel run, where the process is carried out without waiting between any stages of the process, was performed. TAT was obtained. Then, the team repeated the process for batch after batch to obtain the best possible TAT.
To determine a machine/section-wise load, the team needed to determine the number of tests expected per day in each section. This required estimations as follows:
- Number of patients per day
- Samples per patient
- Tests per sample per machine or per station
- Reports per patient
Biochemistry Example
Armed with this data, the green channel run was first piloted and then standardized in each section. For this article’s purposes, consider the example of biochemistry (a category of tests) with the maximum number of tests.
The process and the load – patients, samples per patients, tests per sample and the time per stage – are summarized in Table 5.
- Samples per patient = 2
- Tests per sample = 3
- Tests per patient = 6
- Time per test = 27 seconds
| Table 5: Biochemistry Process and Stage Times | ||||
| Process Map | Biochemistry | Op Unit | Time (Seconds) | Seconds/Sample |
| Accession (prepping a tray of samples to send to lab for processing – paperwork and arrangement) | Smallest unit sample (1 sample test tube) | 16 samples | 900 | <56.25 |
| Centrifuge | 16 samples | 16 samples | 900 | 56.25 |
| Biochemistry Testing | Test | 16 tests/sample | 432 | 27 (equivalent to 400 tests/hour) |
| Report | Patient | Up to 16 samples/patient | 900 | <56.25 |
| Approval | Patient | Up to 16 samples/patient | 900 | <56.25 |
The centrifuge was the bottleneck and was the pace setter for the biochemistry process. It holds only 16 samples at a time and this was, therefore, selected as the minimum batch size. Loading it with minimum interruption provided the maximum output and last TAT.
The process was run, timed and studied in several trial runs with improvements built in and tested in each run. The results of two trials are detailed here.
First Trial
- Start: Load into centrifuge
- Finish: Report approved
- Samples: 17
- Tests: 37
The results are summarized in Table 6.
| Table 6: Trial of Biochemistry Flow Run | ||
| Step | Actual Time (Minutes) | Ideal Time (Minutes) |
| Centrifuge in | 12:13 | 12:13 |
| Centrifuge out | 12:23 | 12:23 |
| Into machine | 12:23 | 12:23 |
| Processing (37 tests at 9 secs/test) | 12:29 | |
| Report check start | 12:30 | |
| Report approve start | 12:31 | |
| Report check finished | 12:36 | |
| Report approve finished | 13:32 | 12:37 |
| TAT | 79 | 24 |
| Expected | 30 | |
Against the ideal time of 24 minutes and expected time of 30 minutes, the actual time achieved was 79 minutes.
Avoidable reasons for the long actual time were:
- Manual barcoding took 12 minutes. (Look for details on addressing this issue in the final part of this article next week.)
- Waiting to be loaded into the analyzer that was avoidable in full flow took 10 minutes.
The achievable cycle time was 57 minutes.
Second Trial
- Start: From sample repeat trial
- Finish: Biochemistry report approved
- Start time: 8:40 am
- End time: Peak sample repeat trial time over by 12:30 p.m.
The results are summarized in Table 7.
| Table 7: Second Trial of Biochemistry Flow Run | |||||
| Step Number | Center Number | In Time | Samples | To Biochemistry | Number of Samples |
| 1 | 1,2 | 08:40 | 42 | 08:55 | 42 |
| 2 | 09:05 | 30 | 09:10 | 30 | |
| 3 | 3,4 | 10:15 | 190 | 10:20 | 16 |
| 10:35 | 66 | ||||
| 10:47 | 53 | ||||
| 10:58 | 51 | ||||
| 11:01 | 12 | ||||
| 11:06 | 1 | ||||
| 11:08 | 1 | ||||
| 4 | 5 | 10:32 | 65 | 11:01 | 55 |
| 11:06 | 10 | ||||
| 5 | 6 | 11:10 | 57 | 12:00 | 57 |
| 6 | 7,8 | 11:32 | 42 | 11:42 | 42 |
| 7 | 7,8 | 12:25 | 22 | 12:33 | 22 |
The run was completed by 1:30 p.m. With the peak time load processed early, the rest of the tests running in the afternoon were easy to manage.
Several improvements were implemented that had significant results.
- Pre-trial process in accession: The first 42 samples were loaded into trays of 16 samples per tray. When one tray was loaded, the second tray was started. The process was to pass on the trays to biochemistry when all samples were loaded.
- A small but significant change in work practice was made with one loaded tray being promptly passed on so that the centrifuge could start as the next tray was loaded. The bottleneck was able to begin running as soon as possible and then it was kept running.
- There was a break between 9:10 a.m. and 10:15 a.m. due to a slipup from the Center related to transferring the samples. This break was removed and the centrifuge machine was able to keep running.
Third Trial
- Number of samples = 16
- Number of tests = 97
The results are shown in Table 8.
| Table 8: Third Trial of Biochemistry Flow Run | |||
| Trial Run for 16 Tubes | Start Time | Time Lost (Minutes) | Comments |
| Sample ready for centrifuge | 10:33 | 4 | · 15 red caps and 1 yellow cap (identifiers for types of sample tubes), which is taller and cannot balance in the centrifuge
· Yellow cap changed for red cap |
| Centrifuge start | 10:37 | ||
| Centrifuge end | 10:47 | 6 | · Worklist not available and had to be retrieved from the accession
· Reference was required to determine tests that cannot be completed by the machine · 1 tube needed electrolyte (potassium) |
| Loaded in the machine | 10:53 | ||
| Barcode reading and ready for test | 10:58 | ||
| Test started in machine | 11:03 | ||
| First tube ready | 11:06 | ||
| Test completed (for all 16 tubes) | 11:40 | ||
| Report review start | 11:40 | 24 | |
| Status changed to completed | 11:50 | Decimal values were added to whole number results (e.g., 14.00 instead of 14) | |
| Approval completed by doctor | 12:00 | 20 | |
| Total time taken | 1:27 | ||
When it came to the time lost in various tasks in the process, there were a couple of areas of concern that were quickly addressed.
- A yellow cap, which was a different height than the others and thus did not balance in the centrifuge, was changed to a red cap so that it could go with the other tests at the same time.
- The delays due to report review and completion were addressed by explaining to the doctors how they needed to fit into the overall process flow.
Addressing of these types of problems continued over several cycles. Two other examples were:
- Tubes of different sizes in one batch or partial batches were sent to the centrifuge. To keep tests moving, balancing dummy tubes were kept at the centrifuge to fill the extra slots as needed.
- The trays from accession to centrifuge could carry 20 tubes, which meant that 16 were loaded while 4 (then 8 and 12) had to wait for the next batch. New trays holding 16 tubes per tray rather than 20 were bought and then to maintain the even flow of tubes.
The above trials and improvements were spread over three days. A summary of the results follows.
- Day 1: No work in process (WIP). Work was finished at 5:30 p.m., unlike before the project when some samples had to be processed overnight (maximum TAT = 4 hours).
- Day 2: No WIP at 5:30 p.m.
The head of the laboratory shared a great compliment: “I used to chase biochemistry; now I have to chase other departments!”
The Ultimate Test – Saturday
Saturday mornings always presented peak load challenges as this tended to be the day of the week with the greatest number of patients. The first Saturday using the new process flow system produced the results shown in Table 9.
| Table 9: Saturday Biochemistry Processing in Flow | |
| Batch | Starting at Minute |
| Batch 1 | 30 |
| Batch 2 | 45 |
| Batch 3 | 60 |
| Batch 4 | 75 |
| Batch 5 | 90 |
| Batch 6 | 105 |
| Batch 7 | 120 |
| Batch 8 | 135 |
| Batch 9 | 150 |
| Batch 10 | 165 |
The efficiency of processing was more than 90 percent.
- Tubes = 160
- Reports = 154
- Start: 9:40 a.m.
- Finish: 12:30 p.m.
- Time to completion: 182 minutes
- Ideal time: 165 minutes
By 1:00 p.m. the peak load had been processed and the worst was over.
Conclusion
The key objective – having 100 percent of the samples received in the morning getting their reports by the end of the same day – was achieved. Approximately 70 percent of the afternoon samples (fewer in number than what was received mornings) were completed by 7 p.m. The reports for those samples were ready to be given to patients by late evening.
Additionally, the revised process made the work easier, easily absorbing peak loads while dramatically reducing the overtime of staff and releasing capacity during the afternoons for additional samples to be processed.
The control chart was found to not be required and was, therefore, discontinued.
| Appendix 2.2: Arrival Pattern of Samples for Testing | ||||
| Blood Samples | ||||
| February 10 | February 11 | February 12 | February 13 | |
| 7:00 a.m. | 8 | 2 | 11 | 20 |
| 8:00 a.m. | 6 | 4 | 30 | 18 |
| 9:00 a.m. | 56 | 29 | 25 | 40 |
| 10:00 a.m. | 52 | 89 | 68 | 57 |
| 11:00 a.m. | 65 | 49 | 44 | 57 |
| 12:00 p.m. | 103 | 84 | 38 | 84 |
| 1:00 p.m. | 99 | 46 | 15 | 76 |
| 2:00 p.m. | 36 | 72 | 42 | 39 |
| 3:00 p.m. | 15 | 51 | 8 | 37 |
| 4:00 p.m. | 18 | 12 | 2 | 33 |
| 5:00 p.m. | 21 | 13 | 2 | 14 |
| 6:00 p.m. | 25 | 15 | 4 | 30 |
| 7:00 p.m. | 19 | 16 | 0 | 10 |
| 8:00 p.m. | 6 | 4 | 1 | 17 |
| 9:00 p.m. | 5 | 5 | 0 | 8 |
| 10:00 p.m. | 11 | 4 | 0 | 9 |
| 11:00 p.m. | 2 | 0 | 0 | 10 |
| 12:00 a.m. | 2 | 0 | 2 | 2 |
| 1:00 a.m. | 0 | 0 | 0 | 2 |
| 2:00 a.m. | 0 | 0 | 2 | 0 |
| 3:00 a.m. | 0 | 0 | 0 | 0 |
| 4:00 a.m. | 0 | 0 | 0 | 0 |
| 5:00 a.m. | 3 | 6 | 0 | 0 |
| 6:00 a.m. | 1 | 11 | 0 | 14 |
| Total | 553 | 512 | 293 | 578 |
| Ave | 23 | 21 | 12 | 24 |
| Appendix 2.3: Arrival Pattern of Samples for Testing | ||||
| Urine Samples | ||||
| February 10 | February 11 | February 12 | February 13 | |
| 7:00 a.m. | 2 | 1 | 4 | 6 |
| 8:00 a.m. | 2 | 1 | 10 | 6 |
| 9:00 a.m. | 18 | 9 | 8 | 13 |
| 10:00 a.m. | 16 | 28 | 22 | 18 |
| 11:00 a.m. | 20 | 15 | 14 | 18 |
| 12:00 p.m. | 33 | 27 | 12 | 26 |
| 1:00 p.m. | 31 | 14 | 5 | 24 |
| 2:00 p.m. | 12 | 23 | 13 | 12 |
| 3:00 p.m. | 5 | 16 | 2 | 12 |
| 4:00 p.m. | 6 | 4 | 0 | 11 |
| 5:00 p.m. | 7 | 4 | 0 | 5 |
| 6:00 p.m. | 8 | 5 | 1 | 9 |
| 7:00 p.m. | 6 | 5 | 0 | 3 |
| 8:00 p.m. | 2 | 1 | 0 | 6 |
| 9:00 p.m. | 1 | 2 | 0 | 2 |
| 10:00 p.m. | 4 | 1 | 0 | 3 |
| 11:00 p.m. | 0 | 0 | 0 | 3 |
| 12:00 a.m. | 1 | 0 | 1 | 0 |
| 1:00 a.m. | 0 | 0 | 0 | 1 |
| 2:00 a.m. | 0 | 0 | 0 | 0 |
| 3:00 a.m. | 0 | 0 | 0 | 0 |
| 4:00 a.m. | 0 | 0 | 0 | 0 |
| 5:00 a.m. | 1 | 2 | 0 | 0 |
| 6:00 a.m. | 0 | 4 | 0 | 5 |
| Total | 175 | 162 | 93 | 182 |